Accenture creates a regulatory document authoring solution using AWS generative AI services
AWS Machine Learning - AI
FEBRUARY 6, 2024
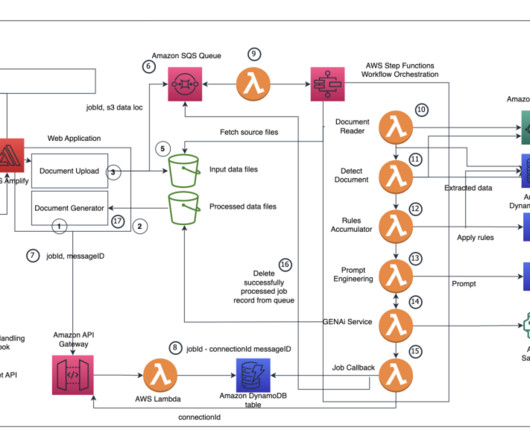
A key part of the submission process is authoring regulatory documents like the Common Technical Document (CTD), a comprehensive standard formatted document for submitting applications, amendments, supplements, and reports to the FDA. The React application uses the Amplify authentication library to detect whether the user is authenticated.








































Let's personalize your content